What Are Electrolytes and Nonelectrolytes? Examples and Differences
2026-04-29
234
Catalog
Electrolytes: Properties and Behavior
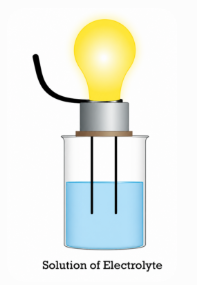
Figure 2. Electrical Conductivity of an Electrolyte Solution
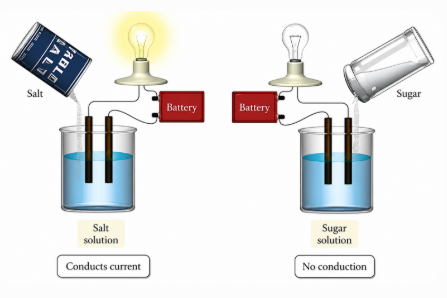
Electrolytes are substances that dissolve in water and produce ions, making their solutions electrically conductive and chemically active.
1. Ion Formation and Dissociation
When electrolytes dissolve, they split into cations and anions. The degree of separation determines their strength:
• Strong electrolytes fully dissociate, producing many ions
• Weak electrolytes partially dissociate, producing fewer ions
This affects both conductivity and reactivity.
Example
In the human body, electrolyte levels influence nerve signals and heart rhythm. In batteries, efficient ion movement improves energy transfer.
2. Electrical Conductivity
Electrolytes conduct electricity because ions carry charge. Ion movement creates electric current, and conductivity increases with the number of ions present.
Example
Seawater conducts electricity well due to its high ion concentration, while pure water has very low conductivity. Electrolyte solutions are also used in electroplating to coat metals.
3. Chemical Reactivity
Electrolytes are reactive in solution because ions can easily interact. This allows processes such as:
• Precipitation reactions
• Acid-base neutralization
• Redox reactions
Example
Mixing silver nitrate and sodium chloride forms a white precipitate of silver chloride. This type of reaction is used in water treatment and analysis.
4. Nature and Composition
Electrolytes are typically:
• Salts (e.g., sodium chloride)
• Acids (e.g., hydrochloric acid)
• Bases (e.g., sodium hydroxide)
They are usually ionic or strongly polar, allowing water to separate them into ions.
5. Solubility and Performance
For effective performance, electrolytes must dissolve well and produce many ions.
Limitation
Some substances dissociate but have low solubility. For example, silver chloride forms ions but dissolves very little, so it has low conductivity.
Nonelectrolytes: Properties and Behavior
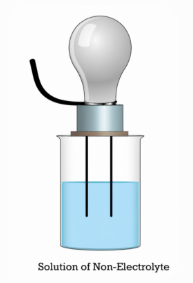
Figure 3. Non-Electrolyte Solution does not Conduct Electricity
Nonelectrolytes differ because they do not form ions in solution. This limits their conductivity and ionic reactivity.
1. No Ion Formation
When nonelectrolytes dissolve, they remain as neutral molecules. No charged particles are produced.
2. Lack of Electrical Conductivity
Since no ions are formed, nonelectrolytes do not conduct electricity. Even with applied voltage, no current flows.
Example
Sugar dissolved in water does not conduct electricity, while saltwater does. The difference is ion formation, not solubility.
3. Limited Chemical Reactivity
Nonelectrolytes do not participate in ionic reactions. Instead, they are involved in:
• Molecular reactions
• Biochemical processes
• Energy-related reactions
Example
Glucose does not form ions but is important for energy production in cells.
4. Nature and Composition
Most nonelectrolytes are covalent compounds, often organic, such as:
• Sugars
• Alcohols
• Urea
They dissolve without breaking into ions.
5. Solubility Without Conductivity
Many nonelectrolytes dissolve in water but remain neutral. This shows that solubility alone does not guarantee conductivity.
Electrolytes vs Nonelectrolytes
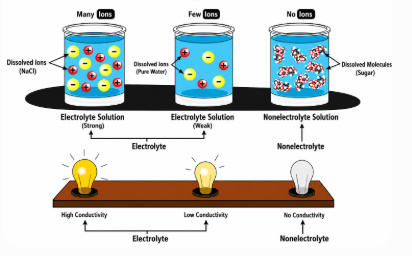
Figure 4. Electrolytes vs Nonelectrolytes Comparison
|
Basis |
Electrolytes |
Nonelectrolytes |
|
Ion Formation |
Forms ions |
No ion formation |
|
Conductivity |
Conduct electricity |
Do not conduct |
|
Particles in Solution |
Ions |
Neutral molecules |
|
Bond Type |
Ionic or polar covalent |
Covalent |
|
Reactivity |
Ionic reactions |
Molecular reactions |
|
Examples |
NaCl, HCl, KOH |
Glucose, ethanol |
Practical Uses and Importance
Understanding these substances has practical applications.
Health and Medicine
Electrolytes regulate hydration, nerve impulses, muscle contraction, and heart rhythm. When electrolyte levels are imbalanced, it can lead to fatigue, cramps, or even serious health issues.
Energy and Technology
Electrolytes are used in batteries, fuel cells, and other electrochemical devices. For example, lithium-ion batteries rely on the movement of ions to generate and store energy, allowing them to power devices efficiently.
Chemistry and Industry
• Electrolytes enable:
Electrolytes are used in chemical reactions in solution, metal extraction and purification, and electrolysis processes.
• Nonelectrolytes are important in:
Nonelectrolytes are used in food and pharmaceuticals, help maintain chemical stability, and support controlled reactions.
Common Misconceptions About Electrolytes and Nonelectrolytes
All soluble substances conduct electricity
Conductivity depends on ion formation, not just solubility.
Strong electrolytes are only strong acids
Strong electrolytes also include strong bases and soluble salts.
Weak electrolytes do not conduct electricity
They do conduct, but less efficiently because they produce fewer ions.
Nonelectrolytes do not dissolve in water
Many dissolve, but they remain as neutral molecules.
Non-conductive means insoluble
A substance can dissolve in water and still not conduct electricity if it does not form ions.
Conclusion
The main difference between electrolytes and nonelectrolytes is ion formation. This simple idea explains their role in conductivity, reactions, and practical applications. Once you understand it, many chemical processes become easier to understand.
 ABOUT US
Customer satisfaction every time. Mutual trust and common interests.
ABOUT US
Customer satisfaction every time. Mutual trust and common interests.
function test. The highest cost-effective products and the best service is our eternal commitment.
Hot Article
- LM358 Dual Operational Amplifier Comprehensive Guide: Pinouts, Circuit Diagrams, Equivalents, Useful Examples
- Are CR2032 and CR2016 Interchangeable?
- Understanding the Differences ESP32 and ESP32-S3 Technical and Performance Analysis
- Choosing the Right Battery: A Guide to AG4, LR626, LR66, 177/376/377, SR626, and SR626SW Equivalents
- BC547 Transistor Basics: Pinout, Application Circuits, Alternative/Complementary Models
- NPN vs. PNP: What's the Difference?
- esp32 vs stm32: which microcontroller is better for you?
- What Is a MOSFET and How It Works?
- Electrical Relay Basic: Working Operation, Types and Uses
- PNP Transistors: Structure, Working Principle and Application
 Alphanumeric Codes: How They Help Your Devices Work
Alphanumeric Codes: How They Help Your Devices Work
2026-04-29
 RS-232 vs RS-485: Which Is Better for Your System
RS-232 vs RS-485: Which Is Better for Your System
2026-04-29
Frequently Asked Questions [FAQ]
1. How does the degree of dissociation affect electrolyte strength and conductivity?
The more a substance dissociates into ions, the stronger the electrolyte and the higher its conductivity. Full dissociation produces many charge carriers, while partial dissociation limits current flow.
2. Why can a strong electrolyte still show low conductivity in some cases?
If a strong electrolyte has low solubility, it produces fewer ions in solution. This reduces conductivity even though it fully dissociates.
3. How does the type of chemical bond influence electrolyte behavior?
Ionic and polar covalent compounds tend to form ions in water, making them electrolytes. Nonpolar covalent compounds usually remain intact, so they act as nonelectrolytes.
4. How is electrolyte behavior related to acid-base reactions?
Electrolytes provide ions such as H⁺ and OH⁻, which drive acid-base reactions. The strength of the electrolyte affects how completely these reactions occur.
5. How does ion concentration influence electrochemical processes like batteries?
Higher ion concentration improves charge transfer between electrodes, increasing efficiency and performance in devices like batteries and fuel cells.
6. How does electrolyte imbalance affect biological systems?
Imbalance can disrupt nerve signals, muscle contraction, and heart rhythm, leading to symptoms like fatigue, cramps, or more serious health issues.
7. Why is understanding electrolytes important in industrial processes?
Electrolytes enable controlled chemical reactions, metal extraction, and electrolysis, making them important in manufacturing and material processing.
Hot Part Number
 C0402X5R1C151M020BC
C0402X5R1C151M020BC GQM1885C1H270GB01D
GQM1885C1H270GB01D CC0201KRX7R7BB471
CC0201KRX7R7BB471 CL21B474KBFNFNG
CL21B474KBFNFNG LD053A472FAB2A
LD053A472FAB2A GRM1555C1HR60BZ01D
GRM1555C1HR60BZ01D GRM1556P1H2R4CZ01D
GRM1556P1H2R4CZ01D C2012X5R1A105M/0.85
C2012X5R1A105M/0.85 1.5KE16A-E3/54
1.5KE16A-E3/54 CS4362A-CQZ
CS4362A-CQZ
- MAX13223EEUP+
- JS28F640P33BF70A
- RT1206DRD073K3L
- MC9S12UF32PUE
- BSM100GB120DN2E
- VI-810218
- UCC2813PW-0
- MC74LCX14DT
- LT3518IUF#PBF
- T491C335M035AT4380
- DAC104S085CISD/NOPB
- GS3471-IBE3
- AK2346-E2
- AUIPS6031RTR
- BSS138NH6327
- CAT22C10WI-20
- CX90220-11Z
- FFPF06F20DN
- HD64F7058F80V
- LMX2434TM
- LSI53C895-208QFP
- LTC1323CS
- MB87F3061P
- MC145574DW
- SA586RQI-V2.0
- SM8952AL25J
- SPDA1030A1-010B-HL09K
- XC17512LJC
- AN16077AA
- HYB39S128160CT-7
- LR38668Y
- SSC2571FOA
- STC8H1K24-36I-LQFP32
- XC18V04VQG44I
- SC8542VTX600K
- CXD4116-1
- M7107B-B1AG
- LMZ10501SIL
- C2012X7R1C225KB